An inventory item
which is consumed as a component in the Additive manufacturing process can have
reference items against it.
Examples of component
inventory items include:
- purchased TPNs e.g. Baxter Olimel/PeriOlimel 3-compartment TPN bag, SmofKabiven electrolyte/electrolyte
free TPN bags
- concentrated
electrolyte for injection e.g. calcium chloride 10% vial, potassium acetate
490mg/mL vial
- infusion
solutions e.g. Hartmann’s infusion bag, sodium chloride 0.45% infusion bag
Reference items describes
the:
- constituents
that make up the inventory product such as amino acid/glucose & other
carbohydrates/lipid/electrolytes/nitrogen content; and
- chemical
& physical properties of the inventory product such as energy content,
osmolality, osmolality and pH
The Reference items
will appear on the printed Additive Label:

For each Reference
Item, the value is the sum total of all components multiplied by the final
volume
e.g. if the
components were NaCl and NaAcetate, the total Na+ will be inclusive of the Na+
of NaCl and NaAcetate multiplied by the final volume.
Setup:
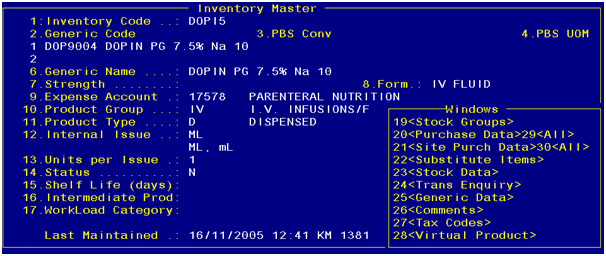
Inventory
code has an associated Generic code. This map is maintained in the Inventory
Master screen.
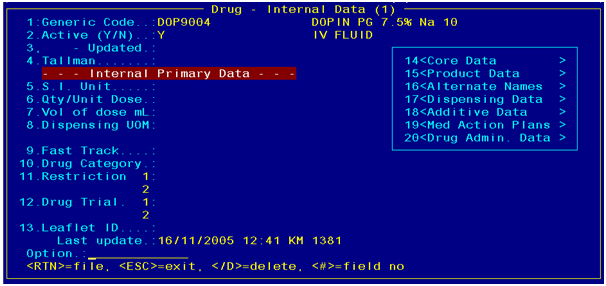
The generic code has additional properties, which are used to
describe the drug. This includes additive properties such as reference items.
This relationship can be maintained in the DRUGS2 screen.
To access this screen:
Dispense
8. Reference File Maintenance
13. Drugs & Related Files - Internal User Data
1. Drug File - Internal Data
OR
zoom [DRUGS2
The reference items data sits in the Additives Data sub-screen
(field 18).
Each
reference item’s values are listed in field 5 and 6. The value in field 6
represents the qty/mL.
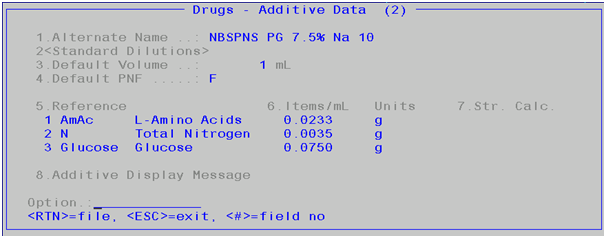
Field 5 is a lookup. The reference item listed in this
lookup and units that appear in the screen is maintained in the “Additives
Reference Item” screen.
To access this screen:
Dispense
8. Reference File Maintenance
13. Drugs & Related Files-Internal User Data
5. Drug Referenced
Item
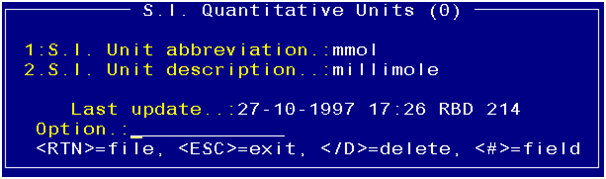
The reference unit (field 3) is a lookup. This value is
maintained in the “S.I. Quantitative Units” screen.
To access this screen:
Dispense
8. Reference File Maintenance
12. Drugs & Related Files-External Core Data
6. Drug S.I. Measure
Unit